The New Food Police
Who will keep our dinner safe?
You wouldn’t want Michael Taylor’s job.
As the Food and Drug Administration’s Deputy Commissioner for Foods and Veterinary Medicine, Taylor bears responsibility for almost all of the food consumed in the U.S. According to the Centers for Disease Control and Prevention, 48 million people here – roughly one in six – contract a foodborne illness each year. Of these, 128,000 will be hospitalized; 3,000 will die. When food makes people sick, Taylor’s agency is a handy fall guy: Why did they let this happen?
For decades, the FDA has been under fire for failing to prevent contagions, for focusing on pharmaceuticals at the expense of food, for inconsistent safety standards. The agency has been criticized on the one hand for weak enforcement, on the other for bullying small producers. “The FDA is no friend of the farmer,” says Pete Kennedy, president of the Farm-to-Consumer Legal Defense Fund.
Our global food systems have grown incredibly complex and interconnected. Massive food processors can quickly distribute tainted goods around the world. When infected salad can cross international borders in mere hours, when tainted peanut paste can end up in 2,000 different products, someone needs to ensure food’s basic safety. Despite being chronically underfunded and understaffed, the FDA has found itself shouldering that burden – worldwide. “We at the FDA have a global food mission,” says Taylor.
But are they up to the challenge?
Ensuring food safety has always been an FDA mandate, but its approach has traditionally been conservative. Lab scientists would spend years determining what was and wasn’t okay to consume. After something was determined to be iffy – like the infamous red dye #2 – it was yanked from the shelves. “We’ve always taken a very reactive approach,” says Taylor.
Then in 1993, the food safety landscape changed forever. The fastfood chain Jack in the Box served E. coli-tainted hamburgers at 73 of its West Coast stores, leaving a grim tally: more than 700 sick and 178 left with permanent kidney and brain damage, most of them younger than 10. Four children died.
“This was the moment when we collectively said: ‘Oh look, our industrialized food system could harm us,’ ” says Diana Winters, professor of food and drug law at Indiana University’s McKinney law school. “Regulators picked up on this … that’s when the USDA started regulating E. coli.”
Over the last century, there’d been a shift toward large, centralized food distribution systems. With that came a heightened potential for quick-moving, far-reaching disease. The danger was emerging, and regulators had not yet caught up. Shortly after the Jack in the Box incident, Taylor (then with the USDA) gave a prescient speech to the American Meat Institute. “There is a gap in our system,” he said. “The fact is we do not deal directly enough and scientifically enough with the microbial pathogens that can make people sick.”
In the two decades following Taylor’s speech, microbial pathogens have gained even more notoriety. In addition, the increasingly global food market means that any problem overseas can be a problem for the FDA – and the problems are legion. Salmonella alone kills over 370,000 people each year around the world. India – the sixth-largest food exporter to the U.S. – is riddled with food-safety issues. China is racked with scandal after scandal: expired meat, high lead levels in baby cereal, tainted milk. Europe has its share of issues as well – in 2011, more than 3,000 Europeans were sickened by sprouts tainted with E. coli.
In the U.S., half of all fresh fruit is now imported. About 90 percent of our seafood comes from abroad. The volume of U.S. food imports has increased almost every year over the past decade. The FDA now devotes half of its inspection staff to imports, many of them from China. The recent mega-merger between giant American meat company Smithfield Foods and China’s Shuanghui International is just one sign of our growing connectivity.
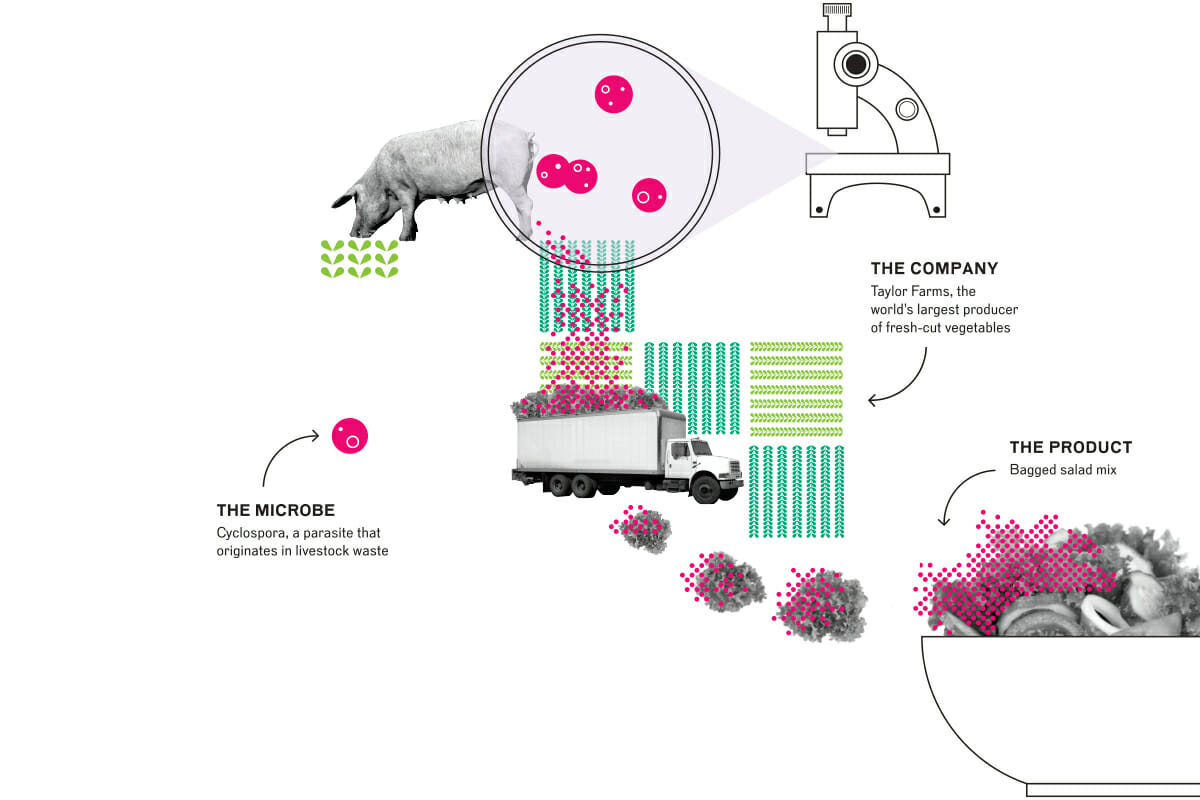
Take, for instance, a large outbreak of cyclosporiasis in 2013. The pathogen that caused part of this outbreak was traced back to bagged salad mix grown in Mexico by the massive produce company Taylor Farms. In a very small window of time (greens have a short shelf life), the salad was linked to a larger outbreak across 25 U.S. states that sickened at least 631 people. The cause was never officially determined, but evidence pointed to hand-washing issues.
After reviewing the case, the FDA made a handful of suggestions for improving the farms’ safety conditions. But, as has long been the case, it was more of a “best practice” guideline than an enforceable mandate.
This kind of large-scale contagion has created a different approach to foodborne pathogens, according to Melinda Wilkins, director of the Master’s program in Food Safety at Michigan State University. Wilkins says that in the past, public health officials perceived food poisoning as “event-based.” For instance, someone brings a bad bowl of potato salad to a potluck, or a restaurant serves an iffy batch of oysters one night. Fifteen out of 25 people at the event get sick, the cause is isolated, case closed.
But when lettuce harvested in Mexico on Saturday can be sold in 25 states by Sunday, there needs to be a different approach – not to mention’a sense of urgency – in determining the source. In many ways, it’s the de-localization of our food that has ramped up the scope of foodborne illness. While the U.S. has a better food safety record than much of the world, global trade borders are becoming a lot more open.
The FDA has plenty to contend with here at home. From 2008 to 2009, a salmonella outbreak in peanut products killed nine people. Nearly 2,000 people were sickened in 2010 from salmonella-tainted eggs. Thirty-three people were killed in 2011, victims of Listeria traced to cantaloupe from a Colorado farm. Microbial outbreaks have wreaked havoc in cilantro, chicken, spinach, tomatoes, ground beef, bagged salads and milk.
Many outbreaks involve an oversight that might go unnoticed were it not for the magnitude of our modern food systems. “When you’re working on such a large scale, your mistakes aren’t going to be missed,” says attorney Bill Marler, who represented the victims in civil trials against Jack in the Box. “Suddenly hundreds of people are sick from your cookie dough.”
Considering the risks of this scale, you might think the FDA wouldn’t have the resources to monitor small domestic producers. But just talk to lawyer Jason Foscolo, a self-described “triage guy” for small food businesses: His “Food Law Firm” steps in when regulatory agencies clamp down.
Each week, Foscolo scans a “naughty list” (his phrase) of food businesses that received warning letters, issued recalls or faced some kind of disciplinary action. “You’re not seeing General Mills or Nabisco there,” he says. “This is mostly your small fish company in Boston, your guy making frozen dim sum in Minneapolis.”
When a recall is requested, Foscolo works with regulators to limit its scope. Large food producers can swallow a fairly severe product loss; recall insurance is a given. But what of the little guys who may not be able to afford insurance? “It can absolutely sink a company,” says Foscolo. “I had one client, had to pull ground beef, we’re only talking about a few hundred pounds here … They barely survived.”
In 2010, federal marshals raided a cheesemaker called Estrella Family Creamery in Montesano, Washington, under orders from the FDA. Their entire inventory was seized. Samples of Estrella’s cheese, made from unpasteurized milk, had tested positive for Listeria, as had several parts of their production facility. No one had ever reported an illness due to the company’s products, but food containing Listeria can be very dangerous, even lethal. Estrella, run by a personable couple and beloved by many, unsuccessfully tried to re-open in 2012.
These cases are lightning rods for groups like the Farm-to-Consumer Legal Defense Fund, which obtains counsel for embattled farmers and small food producers. Their issues often revolve around raw milk, the hotly contested commodity that is illegal in some states and cannot be trafficked across state lines. The idea is that consumers should be able to buy from small, local producers – essentially the opposite of where most Americans get their food – without meddling from the federal government. “The FDA has a one-size-fits-all regulatory scheme,” says Kennedy of the FTCLDF. “It’s one thing to crack down on these huge corporations, but consumers should have the freedom to buy from their local farmer.”
[mf_list_sidebar_item]
Wu Heng at the tender age of 27, Wu, who is hailed as a “food safety Warrior” by the Beijing Review, started China’s first comprehensive food-safety database. His crowd-sourced project now includes thousands of violations, helping Chinese activists to demand accountability.
[/mf_list_sidebar_item]
[mf_list_sidebar_item]
Barbara Kowalcyk after her son died of e. coli complications in 2001, statistician Kowalcyk turned her grief into action. she created the Center for foodborne Illness research & Prevention, which uses scientific research to advocate for better food safety standards.
[/mf_list_sidebar_item]
[mf_list_sidebar_item]
Bill Keene the big-bearded epidemiologist/sleuth Keene, responsible for cracking many of the biggest foodborne-illness puzzles, passed away in 2013. the National science foundation honored him with a posthumous lifetime achievement award.
[/mf_list_sidebar_item]
[mf_list_sidebar_item]
Caroline Smith DeWaal as director of food safety at consumer watch-dog Center for science in the Public Interest, deWaal is a force to reckon with. she has testified in front of Congress more than 20 times and frequently advises the Who on food safety issues.
[/mf_list_sidebar_item]
For his part, Marler doesn’t buy the idea that small food producers should be exempt from oversight. “I’m sorry, but it’s your responsibility not to poison people,” he says. “The idea that no one should tell you how to run your business, well that’s getting into ‘Obama wants our guns’ territory.”
But these arguments are more than libertarian-tinged rejections of Big Government. While the FDA argues that raw milk is risky – claiming unpasteurized dairy products are 150 times more likely to cause illness – its advocates point to the numerous foodborne contagions spread by giant agribusiness. There’s something to be said for knowing your local farmer. “It’s about transparency and traceability,” says Kennedy. “You’re never going to eliminate risk, but local food sure has a better track record.”
One thing is clear: Whether your food comes from Mexico or Minnesota, from General Mills or the general store, minimum standards must be ensured. And in the coming years, the FDA has been given the monumental task of overhauling those standards.
The need for new solutions has not gone unnoticed. In 2011, the President signed the Food Safety Modernization Act, or FSMA (pronounced fizzmuh) into law. Taylor calls it the single most important thing to happen to food safety since the start of the country’s modern regulatory system in 1906. FSMA is a sweeping piece of legislation, with mandates that include the establishment of clear produce safety standards, increased importer accountability and partnerships with foreign governments. It grants the FDA an array of new powers, most notably the mandatory recall.
The FDA is now shoring up their standards in virtually every area of food production, from irrigation to compost to hand washing. It will ramp up inspections. It will work with large companies to map out detailed safety plans. No longer consigned to a reactive role – rooting out the cause after food is contaminated – the FDA will now turn its focus on prevention. And to help with this mandate, Congress granted significant enforcement power. The only thing the FDA didn’t receive? Adequate funding.
The agency’s food arm has long borne the reputation of being stretched thin: “This is a chronically underfunded program,” Taylor bluntly admits. Contrast its meager resources with those of the USDA, the agency charged with meat and poultry oversight (roughly 20 percent of our overall food supply). The USDA employs close to 8,000 full-time meat inspectors. By comparison, the FDA employs less than 1,000 investigators for food-safety work that includes inspections – for a nation of 316 million.
The FDA is trying to build partnerships with a handful of foreign regulators so that the U.S. can import with confidence from those countries. Thus far, only New Zealand was deemed up to snuff for this new type of agreement, with Canada soon to follow. New Zealand has demonstrated a world-class safety record, according to Taylor, but even they aren’t exempt from outbreaks. Just this October, more than 100 people there were sickened from an outbreak’of the uncommon pathogen Yersinia pseudotuberculosis. Bagged carrots and lettuce were the likely culprits.
Microbial outbreaks have wreaked havoc in cilantro, chicken, spinach, tomatoes, ground beef, bagged salads and milk.
Other countries, such as Mexico, are doing their best but still struggling to meet U.S. standards. And when it comes to countries whose facilities are especially high-risk, the FDA actually has to send inspectors overseas. Last year they completed 1,400 inspections at foreign facilities, up from 300 the year before. Taylor says half his staff is now working on imports.
To have any hope at effectively safeguarding our food, the FDA will need significant resource allocation. In August, Reps. Rosa DeLauro (D-CT) and Sam Farr (D-CA) sent a letter to the Office of Management and Budget asking why FSMA has yet to be fully funded almost four years after its passage. “If there is an outbreak or an illness related to just one crop,” says Farr, “it could break our trust and sink that crop’s entire industry. Food safety is what provides the public with the confidence needed to keep the market working.”
The FDA would also like the funds to purchase more high-tech equipment for pathogen detection; advances in technology are already helping the agency identify outbreaks. Take a recent case of salmonella-spiked peanut butter. One company’s peanut paste had been used in thousands of products – such as cookies and cracker sandwiches – that were then disseminated nationally. Without modern detection software and tracking databases, it would have been virtually impossible to link all the victims. “There was a time when someone would get food poisoning and think it was an isolated incident,” says Wilkins. “We didn’t have the tools to make connections.”
FSMA could also end up saving money down the road. For instance, forcing importers to ensure the safety of the goods they bring in can be a valuable – if not totally reliable – way of lessening the FDA’s inspection burden. Creating partnerships with foreign regulatory bodies could do the same. And once action plans are set up for various large food producers, they could become more effective at self-policing.
Another key element of FSMA could provide relief for small farmers. The Tester-Hagan Amendment excludes farms with food sales of less than $500,000 a year from FDA regulation. The burden of ensuring safe practices with these producers would fall to state and local authorities. Of course, this leaves room for loose or inconsistent standards – a serious concern for Marler and other safety advocates – but it also frees up the FDA to concentrate on bigger fish.
It should be noted that FSMA is a work in progress. Taylor expects its rules to evolve for years to come. This lets the FDA be more nimble and responsive, to see what works and reject what doesn’t. It can also allow for a dialogue between the agency, consumers and food producers; new amendments come with a public comment period.
Could FSMA herald a new era of responsive regulation? Early signs are positive. “Mike Taylor is doing an admirable job listening to small farmers here,” says Marler. “He changed the rules on washing onions in response to some farmers in the Northwest. And it looks like he’s going to change the rules for cheesemakers very soon.”
That leaves one important group still to be satisfied: consumers. Foodborne illness has become a nightly news staple in the last decade; people are rightly concerned for their safety. A certain amount of media savvy will have to be part of the FDA’s strategy.
“It’s such a tricky balance,” says Winters. “[The FDA] has to listen to what the public thinks they need to stay safe, while also trying to actually protect them from harm.”
Michael Taylor has his work cut out for him.
FOOD DETECTIVES GO HIGH-TECH
Microwave Sterilization
A team of industry, university and military scientists at Washington state University discovered that briefly dunking food underwater and zapping it at a frequency of 915 MHz (stronger than a home microwave) eliminates pathogens and microorganisms that cause spoilage.
Radiation Scanner
Design students in Sweden are working on a countertop food radiation scanner in response to post-Fukushima fears in Japan.
Safety Dipstick
Canadian scientists at McMaster University created a “dipstick” – a 10-centimeter paper strip – that tests for trace amounts of pesticides in food. the technology could be modified to test for e. coli and other foodborne bacteria.
Crop Sampler
Kentucky company Hollison patented a way to test crops for contaminants as they move through the supply chain. rather than testing sporadic samples, this aerosol-based technology scans entire loads of dry goods like wheat, coffee beans and peanuts.
Data Tracking
the CdC has kept a comprehensive database called foodNet since 1996, tracking outbreaks nationwide. In China – where food safety tops consumer concerns – a young writer named Wu Heng (see Heroes of food safety sidebar) started an ambitious crowd-sourced database of food safety incidents.
Follow us

This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License.
Want to republish a Modern Farmer story?
We are happy for Modern Farmer stories to be shared, and encourage you to republish our articles for your audience. When doing so, we ask that you follow these guidelines:
Please credit us and our writers
For the author byline, please use “Author Name, Modern Farmer.” At the top of our stories, if on the web, please include this text and link: “This story was originally published by Modern Farmer.”
Please make sure to include a link back to either our home page or the article URL.
At the bottom of the story, please include the following text:
“Modern Farmer is a nonprofit initiative dedicated to raising awareness and catalyzing action at the intersection of food, agriculture, and society. Read more at <link>Modern Farmer</link>.”
Use our widget
We’d like to be able to track our stories, so we ask that if you republish our content, you do so using our widget (located on the left hand side of the article). The HTML code has a built-in tracker that tells us the data and domain where the story was published, as well as view counts.
Check the image requirements
It’s your responsibility to confirm you're licensed to republish images in our articles. Some images, such as those from commercial providers, don't allow their images to be republished without permission or payment. Copyright terms are generally listed in the image caption and attribution. You are welcome to omit our images or substitute with your own. Charts and interactive graphics follow the same rules.
Don’t change too much. Or, ask us first.
Articles must be republished in their entirety. It’s okay to change references to time (“today” to “yesterday”) or location (“Iowa City, IA” to “here”). But please keep everything else the same.
If you feel strongly that a more material edit needs to be made, get in touch with us at [email protected]. We’re happy to discuss it with the original author, but we must have prior approval for changes before publication.
Special cases
Extracts. You may run the first few lines or paragraphs of the article and then say: “Read the full article at Modern Farmer” with a link back to the original article.
Quotes. You may quote authors provided you include a link back to the article URL.
Translations. These require writer approval. To inquire about translation of a Modern Farmer article, contact us at [email protected]
Signed consent / copyright release forms. These are not required, provided you are following these guidelines.
Print. Articles can be republished in print under these same rules, with the exception that you do not need to include the links.
Tag us
When sharing the story on social media, please tag us using the following: - Twitter (@ModFarm) - Facebook (@ModernFarmerMedia) - Instagram (@modfarm)
Use our content respectfully
Modern Farmer is a nonprofit and as such we share our content for free and in good faith in order to reach new audiences. Respectfully,
No selling ads against our stories. It’s okay to put our stories on pages with ads.
Don’t republish our material wholesale, or automatically; you need to select stories to be republished individually.
You have no rights to sell, license, syndicate, or otherwise represent yourself as the authorized owner of our material to any third parties. This means that you cannot actively publish or submit our work for syndication to third party platforms or apps like Apple News or Google News. We understand that publishers cannot fully control when certain third parties automatically summarize or crawl content from publishers’ own sites.
Keep in touch
We want to hear from you if you love Modern Farmer content, have a collaboration idea, or anything else to share. As a nonprofit outlet, we work in service of our community and are always open to comments, feedback, and ideas. Contact us at [email protected].by Jesse Hirsch, Modern Farmer
December 15, 2014
Modern Farmer Weekly
Solutions Hub
Innovations, ideas and inspiration. Actionable solutions for a resilient food system.
ExploreExplore other topics
Share With Us
We want to hear from Modern Farmer readers who have thoughtful commentary, actionable solutions, or helpful ideas to share.
SubmitNecessary cookies are absolutely essential for the website to function properly. This category only includes cookies that ensures basic functionalities and security features of the website. These cookies do not store any personal information.
Any cookies that may not be particularly necessary for the website to function and are used specifically to collect user personal data via analytics, ads, other embedded contents are termed as non-necessary cookies.